MarketLens
What's Driving Rocket Pharmaceuticals' Latest FDA Review for KRESLADI

Key Takeaways
- Rocket Pharmaceuticals (RCKT) faces a pivotal PDUFA date of March 28, 2026, for its gene therapy KRESLADI, targeting the ultra-rare and deadly Leukocyte Adhesion Deficiency Type 1 (LAD-I).
- The FDA's acceptance of the BLA resubmission, following previous Chemistry, Manufacturing, and Controls (CMC) related Complete Response Letters (CRLs), signals confidence in Rocket's ability to address prior concerns.
- KRESLADI boasts compelling clinical data, including 100% overall survival at 12 months post-infusion and no serious treatment-related adverse events, addressing a severe unmet medical need.
What's Driving Rocket Pharmaceuticals' Latest FDA Review for KRESLADI?
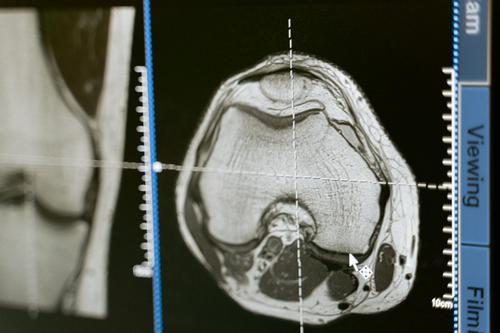
Rocket Pharmaceuticals is at a critical juncture, with the FDA setting a PDUFA date of March 28, 2026, for KRESLADI (marnetegragene autotemcel), its investigational gene therapy for severe Leukocyte Adhesion Deficiency Type 1 (LAD-I). This isn't Rocket's first rodeo with KRESLADI; the journey has been marked by regulatory hurdles, specifically two previous Complete Response Letters (CRLs) from the FDA. Both CRLs, issued in June 2024 and after an initial PDUFA extension in February 2024, centered on requests for additional Chemistry, Manufacturing, and Controls (CMC) information.
The FDA's acceptance of Rocket's BLA resubmission in October 2025 is a significant de-risking event. It signals that the company has likely provided the "limited" additional CMC information the agency required to complete its review. This continued dialogue and the FDA's willingness to re-engage demonstrate a collaborative approach, especially given the high unmet medical need for LAD-I patients. The fact that the FDA confirmed no advisory committee meeting is required further streamlines the review process, suggesting the agency is comfortable with the clinical profile and the resubmitted CMC data.
For a biotech company like Rocket, navigating the intricate regulatory landscape is paramount. The prior delays, while frustrating, underscore the FDA's rigorous standards for gene therapies, particularly concerning manufacturing consistency and quality. The current PDUFA date represents a culmination of these efforts, placing Rocket on the precipice of potentially launching its first commercial product. This milestone could not only provide a life-changing therapy for LAD-I patients but also validate Rocket's lentiviral vector gene therapy platform.
CEO Gaurav Shah emphasized the importance of this therapy, stating that "survival beyond childhood is uncommon" for LAD-I patients and that existing bone marrow transplants carry "substantial morbidity, mortality, and cost." The urgency of the need for an effective, one-time treatment is palpable, adding weight to the FDA's upcoming decision. All eyes are now on March 28, 2026, as Rocket Pharmaceuticals awaits the verdict that could transform its trajectory.
How Strong is the Clinical Data Supporting KRESLADI's Approval?
KRESLADI's path to potential approval is underpinned by exceptionally strong efficacy and safety results from its global Phase 1/2 clinical trial. This data forms the bedrock of Rocket's BLA resubmission and is a primary reason for optimism regarding the upcoming FDA decision. The trial demonstrated a 100% overall survival rate at 12 months post-infusion for all enrolled LAD-I patients, with follow-up extending up to 42 months in some cases. This is a remarkable outcome for a disease where 60-75% of patients typically die before age two without a transplant.
Beyond survival, KRESLADI met all primary and secondary endpoints, showcasing a comprehensive clinical benefit. Patients experienced a significant reduction in severe infections compared to their pre-treatment history, a critical improvement given the life-threatening nature of recurrent infections in LAD-I. Furthermore, the therapy led to the healing of chronic skin lesions and the restoration of wound-healing functions, directly addressing key debilitating symptoms of the disorder. These improvements are not merely statistical; they represent a profound enhancement in the quality of life for these severely affected children.
Safety data from the trial is equally compelling, with KRESLADI being "very well tolerated" and reporting no serious treatment-related adverse events. This clean safety profile is crucial for gene therapies, which often face scrutiny regarding potential off-target effects or severe toxicities. The therapy works by utilizing patient-derived hematopoietic stem cells, genetically modified with a lentiviral vector to deliver a functional ITGB2 gene. This gene is vital for producing the CD18 protein, which facilitates immune cell adhesion and migration to fight infections, effectively correcting the root cause of LAD-I.
The robust clinical package, encompassing both efficacy and safety, positions KRESLADI favorably for approval. The therapy has already received multiple expedited designations, including Regenerative Medicine Advanced Therapy (RMAT), Rare Pediatric, and Fast Track designations in the U.S., as well as PRIME and ATMP designations in the EU. These designations reflect the high unmet need and the promising nature of the clinical data, reinforcing the potential for KRESLADI to become a transformative, one-time treatment for LAD-I.
What Are the Commercial Implications of a KRESLADI Approval?
A positive FDA decision for KRESLADI would be a game-changer for Rocket Pharmaceuticals, marking its transition from a research and development-focused biotech to a commercial-stage company. The immediate financial upside extends beyond direct sales. Upon approval, Rocket would qualify for a Rare Pediatric Disease Priority Review Voucher (PRV). These vouchers are highly valuable assets, often sold to larger pharmaceutical companies for substantial cash payments, historically ranging from $70 million to $350 million. This cash infusion could significantly bolster Rocket's balance sheet, currently holding a market cap of $513.5 million, providing non-dilutive capital to fund its extensive pipeline.
While LAD-I is an ultra-rare disease, affecting a small patient population, gene therapies for such conditions command premium pricing due to their transformative, potentially curative nature and the high cost of alternative treatments like bone marrow transplants. This allows for a significant revenue stream even with a limited patient base. Wall Street analysts project Rocket's revenue to reach $0.2 billion by FY 2029 and $0.4 billion by FY 2030, reflecting the anticipated commercial launch of KRESLADI and other pipeline assets. These estimates, while forward-looking, highlight the market's expectation of KRESLADI's commercial success.
Beyond the direct revenue and PRV, KRESLADI's approval would serve as a powerful validation of Rocket's lentiviral vector platform and its overall gene therapy development capabilities. This validation could attract further investment, facilitate partnerships, and accelerate the development of its other promising programs, including those for Fanconi Anemia, Pyruvate Kinase Deficiency, and Danon Disease. The company's strategic focus on hematology programs, coupled with its cardiovascular pipeline, suggests a diversified approach to rare disease gene therapy.
The commercialization of KRESLADI would establish Rocket's initial market infrastructure, including manufacturing, distribution, and patient support services. This foundational experience will be invaluable as other pipeline candidates mature. For a company with a current P/S ratio of 0.00 (due to no current revenue), the launch of KRESLADI represents the critical first step toward generating meaningful sales and achieving profitability, fundamentally altering its financial profile and investor perception.
What Are the Key Risks and Challenges Facing Rocket Pharmaceuticals?
Despite the strong clinical data and the FDA's acceptance of the BLA resubmission, Rocket Pharmaceuticals faces several inherent risks typical of a clinical-stage biotech company nearing commercialization. The most immediate risk, of course, is the FDA's final decision on March 28, 2026. While the resubmission acceptance is positive, there's always a possibility of unexpected issues, even with "limited" CMC information requests. A third CRL, though less likely now, would be a significant setback, impacting investor confidence and potentially requiring further delays and capital.
Beyond regulatory approval, commercialization presents its own set of challenges. Gene therapies are complex to manufacture, distribute, and administer. Scaling up production to meet potential demand while maintaining stringent quality control is crucial, especially given the prior CMC concerns. The ultra-rare nature of LAD-I means identifying and reaching eligible patients will require specialized commercial efforts, including strong relationships with rare disease centers and patient advocacy groups. Payer reimbursement for high-cost gene therapies can also be a hurdle, requiring robust health economic data and negotiation strategies.
Financially, Rocket Pharmaceuticals is still a pre-revenue company with negative earnings per share (-1.71 TTM). While the potential PRV offers a cash boost, sustained profitability will depend on KRESLADI's market penetration and the successful advancement of its broader pipeline. The company's current cash burn, typical for a biotech in its stage, means it will need to carefully manage its capital, potentially through future equity raises, which could dilute existing shareholders.
Investor sentiment remains volatile for RCKT. The stock currently trades at $4.73, down significantly from its 52-week high of $8.89. While the consensus analyst rating is a "Buy" from 13 analysts, with a median price target of $6.00, there are also "Hold" and "Sell" ratings, indicating a divergence of opinions. Goldman Sachs, for instance, recently maintained a "Sell" rating, albeit with a raised price target to $3.00 from $2.00. This mixed sentiment highlights the speculative nature of investing in biotech companies awaiting major regulatory catalysts.
How Does Rocket Pharmaceuticals Stack Up Against Competitors and the Broader Gene Therapy Market?
Rocket Pharmaceuticals operates within the highly competitive and rapidly evolving gene therapy landscape, but its focus on ultra-rare diseases like LAD-I places it in a niche with limited direct competition. The primary "competitor" for KRESLADI isn't another gene therapy but rather the current standard of care: bone marrow transplant. While effective for some, transplants carry significant risks, including substantial morbidity and mortality, and may not be available in time for all patients due to donor matching challenges. KRESLADI's potential as a one-time, autologous gene therapy (using the patient's own cells) offers a distinct advantage by eliminating donor-related complications and potentially providing a more accessible treatment option.
The broader gene therapy market is experiencing significant innovation and regulatory activity. Recent FDA approvals, even for therapies with mixed clinical data, suggest a growing willingness by the agency to greenlight treatments for severe, unmet needs. This trend could be favorable for Rocket, especially given KRESLADI's strong clinical profile. However, the market is also characterized by intense R&D, with numerous companies vying for breakthroughs in various rare genetic disorders. Rocket's multi-platform approach, leveraging both lentiviral and adeno-associated viral (AAV) vectors, positions it to address a diverse range of diseases, differentiating it from companies focused on a single technology.
From an investment perspective, Rocket's valuation metrics reflect its pre-commercial stage. A negative P/E ratio of -2.36 and a P/S ratio of 0.00 are typical for companies awaiting their first product launch. Its enterprise value (EV) of $460.9 million is slightly below its market cap, indicating a relatively healthy cash position or low debt. The company's current ratio of 6.38 suggests strong short-term liquidity, providing a buffer as it prepares for commercialization.
Analyst consensus points to a "Buy" rating, with a median price target of $6.00, implying an upside from the current $4.73 price. This optimism is likely tied to KRESLADI's potential approval and the value of the associated PRV. However, the wide range of price targets, from a low of $5.00 to a high of $7.00, underscores the inherent uncertainty. Rocket's ability to successfully commercialize KRESLADI and advance its pipeline will be key to realizing these higher valuations and establishing itself as a leader in the gene therapy space.
What Does This Mean for Investors?
For investors, Rocket Pharmaceuticals represents a high-stakes, high-reward opportunity as it approaches the critical PDUFA date for KRESLADI. The FDA's acceptance of the BLA resubmission, coupled with robust Phase 1/2 clinical data showing 100% overall survival and a clean safety profile, significantly de-risks the regulatory pathway. This isn't just about KRESLADI; a successful approval would validate Rocket's entire gene therapy platform and provide a substantial financial boost through a Rare Pediatric Disease Priority Review Voucher.
However, investors must weigh these prospects against the inherent volatility and risks of the biotech sector. The stock's current price of $4.73 reflects some skepticism, trading well below its 52-week high. While the potential for a significant upside exists, particularly if KRESLADI is approved and successfully commercialized, the company still faces challenges in manufacturing scale-up, market access, and managing its cash burn as a pre-revenue entity.
The upcoming PDUFA date on March 28, 2026, is the immediate catalyst. A positive decision could send shares soaring, while an unexpected delay or rejection would likely trigger a sharp decline. Investors should closely monitor any pre-PDUFA communications from the company or the FDA and consider their risk tolerance before making any investment decisions in this speculative, yet potentially transformative, biotech play.
Rocket Pharmaceuticals is on the cusp of a major inflection point. A KRESLADI approval would not only bring a life-saving therapy to children with LAD-I but also establish Rocket as a commercial gene therapy player, potentially unlocking significant long-term value for shareholders. The next few weeks will be pivotal, shaping the company's future and offering a compelling narrative for those willing to embrace the risks of the biotech frontier.
Want deeper research on any stock? Try Kavout Pro for AI-powered analysis, smart signals, and more. Already a member? Add credits to run more research.
Related Articles
Category
You may also like


Why Kiniksa Pharmaceuticals International Stock Raced Nearly 24% Higher Today

Solid Biosciences Announces Receipt of European Commission Orphan Drug Designation for SGT-003 for the Treatment of Duchenne Muscular Dystrophy

BioXcel Therapeutics Preps IGALMI At-Home Launch Ahead of Nov. 14 FDA Label Decision
Breaking News
View All →Featured Articles
Top Headlines
The Stock Market Flashed This Warning Only Once Before. What Comes Next Isn’t Pretty

AMD Likely To Report Higher Q1 Earnings; These Most Accurate Analysts Revise Forecasts Ahead Of Earnings Call

1 Unstoppable Stock to Buy Before It Joins Nvidia, Alphabet, Microsoft, and Apple in the $3 Trillion Club

Meta to expand teen safeguards to 27 EU countries, Facebook safeguards in June